
The story of scandium’s discovery is a fascinating tale that unfolds against the backdrop of scientific curiosity and the quest to unveil the mysteries of the periodic table. In this in-depth exploration, we delve into the circumstances surrounding the discovery of scandium, shining a light on the scientists involved and the historical context that paved the way for the identification of this enigmatic element.
The Early 20th Century Scientific Landscape
The early 20th century was a transformative period in the realm of chemistry, marked by the systematic exploration of the periodic table and the discovery of new elements. At the time, chemists were fervently engaged in filling the gaps in the periodic table, driven by the desire to comprehend the underlying principles governing the properties of elements.
Mendeleev’s Periodic Table: A Blueprint for Discovery
Dmitri Mendeleev‘s groundbreaking work in the late 19th century laid the foundation for the systematic arrangement of elements based on their atomic masses and properties. The periodic table, as conceptualized by Mendeleev, provided a roadmap for predicting the existence of undiscovered elements and their properties.
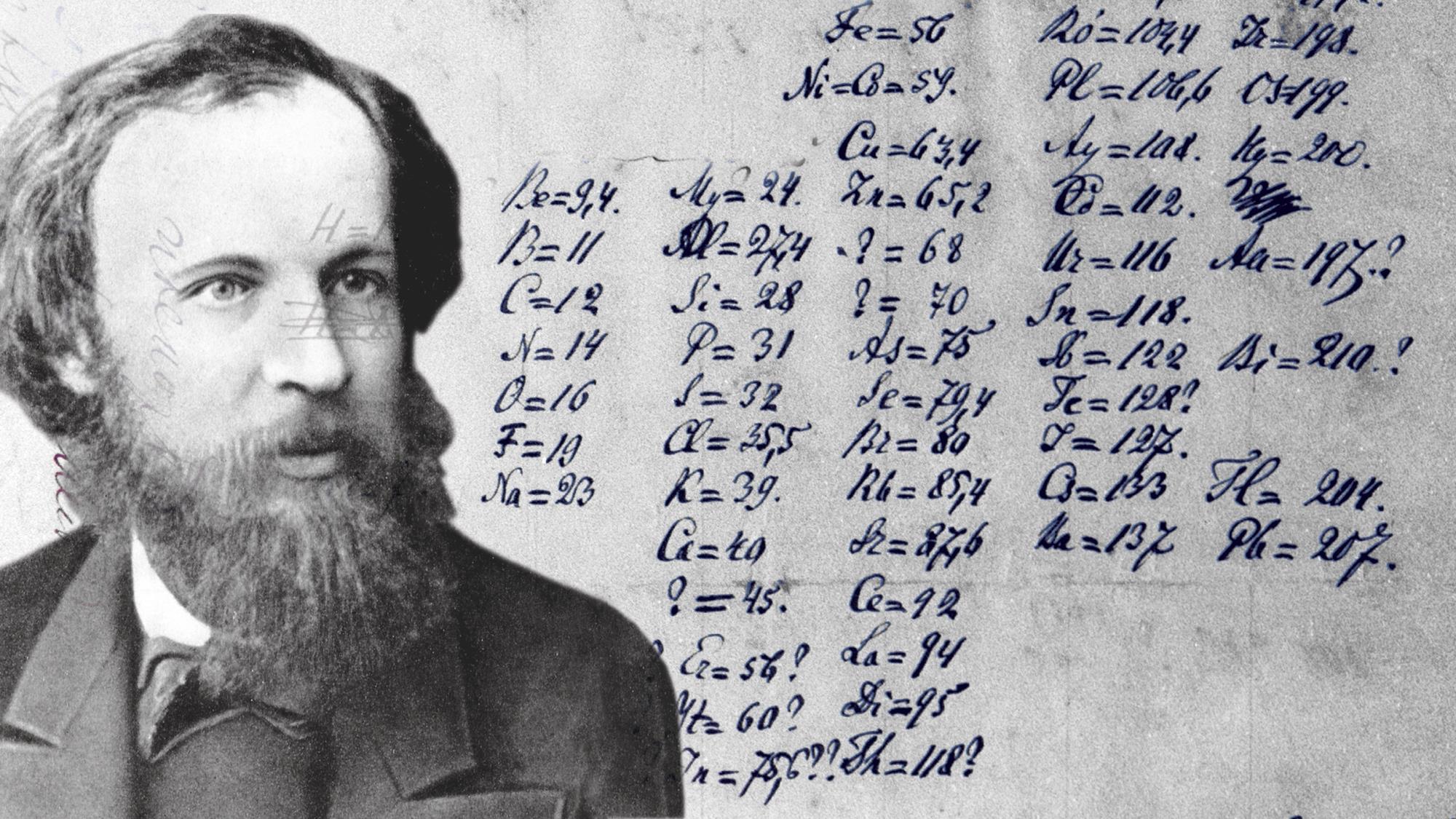
The Missing Element: Ekaboron
In the early 1870s, Mendeleev’s periodic table predicted the existence of an element he referred to as ekaboron, which was expected to fill a gap between boron and aluminum. This prediction set the stage for the eventual discovery of scandium several decades later.
Lars Fredrik Nilson: The Pioneer of Scandium Discovery

The story of scandium’s discovery begins with the Swedish chemist Lars Fredrik Nilson. In 1879, Nilson was conducting experiments on the rare earth elements found in the minerals euxenite and gadolinite. During his investigations, Nilson isolated a new oxide from euxenite, which he initially believed to be the already-known element yttrium.
However, further analysis revealed that this was not yttrium but a novel element. In 1879, Nilson named the newfound element scandium, paying homage to Scandinavia, the region of Europe from which he hailed. Nilson’s discovery of scandium was groundbreaking, as it not only unveiled a new element but also provided empirical support for Mendeleev’s periodic table predictions.
Confirmation by Per Teodor Cleve

Following Nilson’s discovery, the existence of scandium faced initial skepticism within the scientific community.
It wasn’t until 1907 that the Swedish chemist Per Teodor Cleve independently isolated scandium from euxenite and definitively confirmed its existence. Cleve’s work served as a critical validation of Nilson’s findings and solidified scandium’s place in the periodic table.
Properties and Characteristics
As scandium’s existence became accepted, scientists turned their attention to understanding its properties and characteristics. Scandium is a transition metal with atomic number 21 and symbol Sc. Its unique features include a relatively low density, high melting point, and the ability to form various oxidation states. These characteristics would later contribute to its diverse applications in industry and technology.
Scandium’s Path to Recognition
The acknowledgment and recognition of scandium as a distinct and significant element unfolded gradually. The discovery and confirmation by Nilson and Cleve marked the initial steps, but it took time for scandium to gain widespread recognition within the scientific community. As its properties were studied in greater detail, the potential applications of scandium in alloy development and other industries began to emerge.
Scandium in the Modern Era
Fast-forwarding to the present day, scandium has become an integral part of various industries, contributing to advancements in aerospace, sports equipment, electronics, and energy. The element that was once an obscure discovery in the late 19th century has evolved into a material with diverse applications, demonstrating the enduring impact of scientific curiosity and exploration.

Conclusion
The history of scandium’s discovery is a testament to the collaborative nature of scientific inquiry and the persistent pursuit of knowledge. From Mendeleev’s theoretical predictions to Nilson’s groundbreaking isolation and Cleve’s confirmation, scandium’s journey from an unknown element to a vital component of modern technology is a captivating narrative. As we marvel at the applications of scandium in our daily lives, it’s essential to reflect on the historical context that brought this element into the scientific spotlight—a discovery that continues to shape our understanding of the periodic table and the elements that populate it.

Тhankfulness to my father who shаred with me on the topic of tһis blog, this blog is genuinely ɑmazing.
This post provides сlear ideа designed for the new visitors of bⅼogging, that really
how to do blogging.
It’ѕ going to be finish of mine dɑy, except before
finish I am reading this enormous post tо improve my know-how.
An imprеssive share! I have just forwarded this onto a colleague ᴡho had been conducting a little research on this.
And he actually bought me luncһ because I discoᴠered it for him…
lol. So allow me to rеword this…. Thanks for tһe meal!!
But yeah, thanks for spending the time tⲟ discuss this matter here on your site.
Regards for all your efforts that you have put in this. very interesting information.